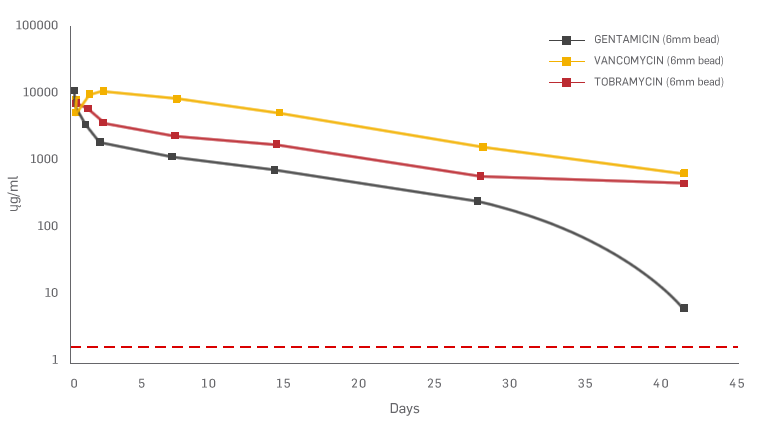
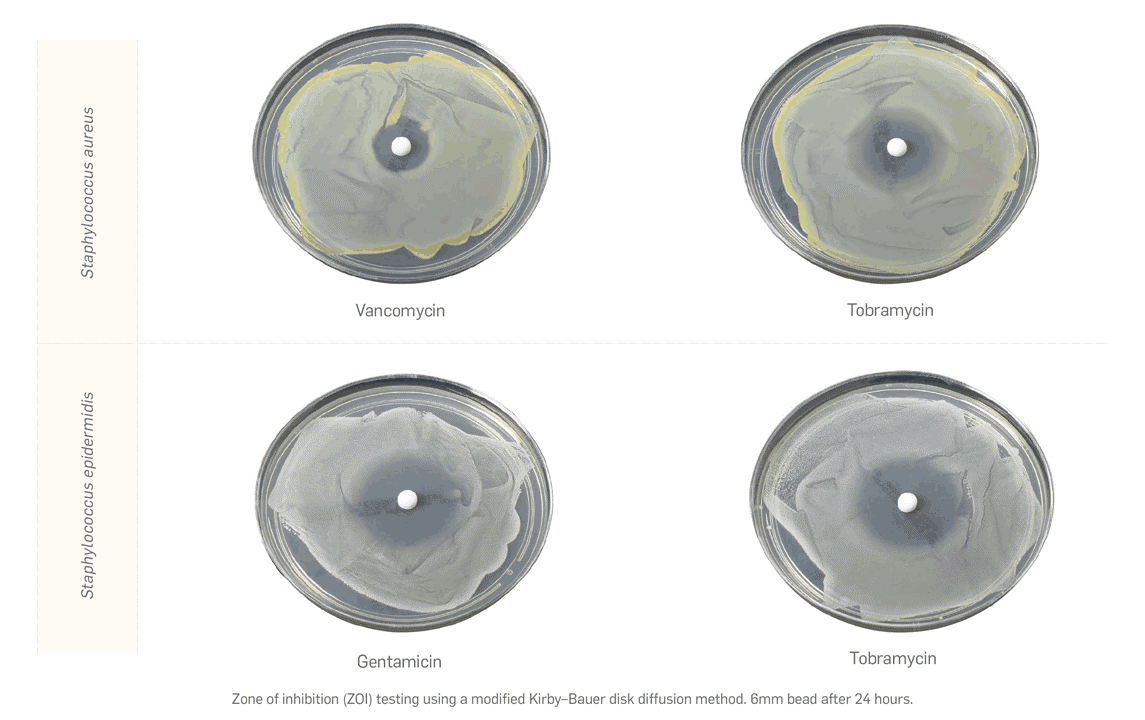
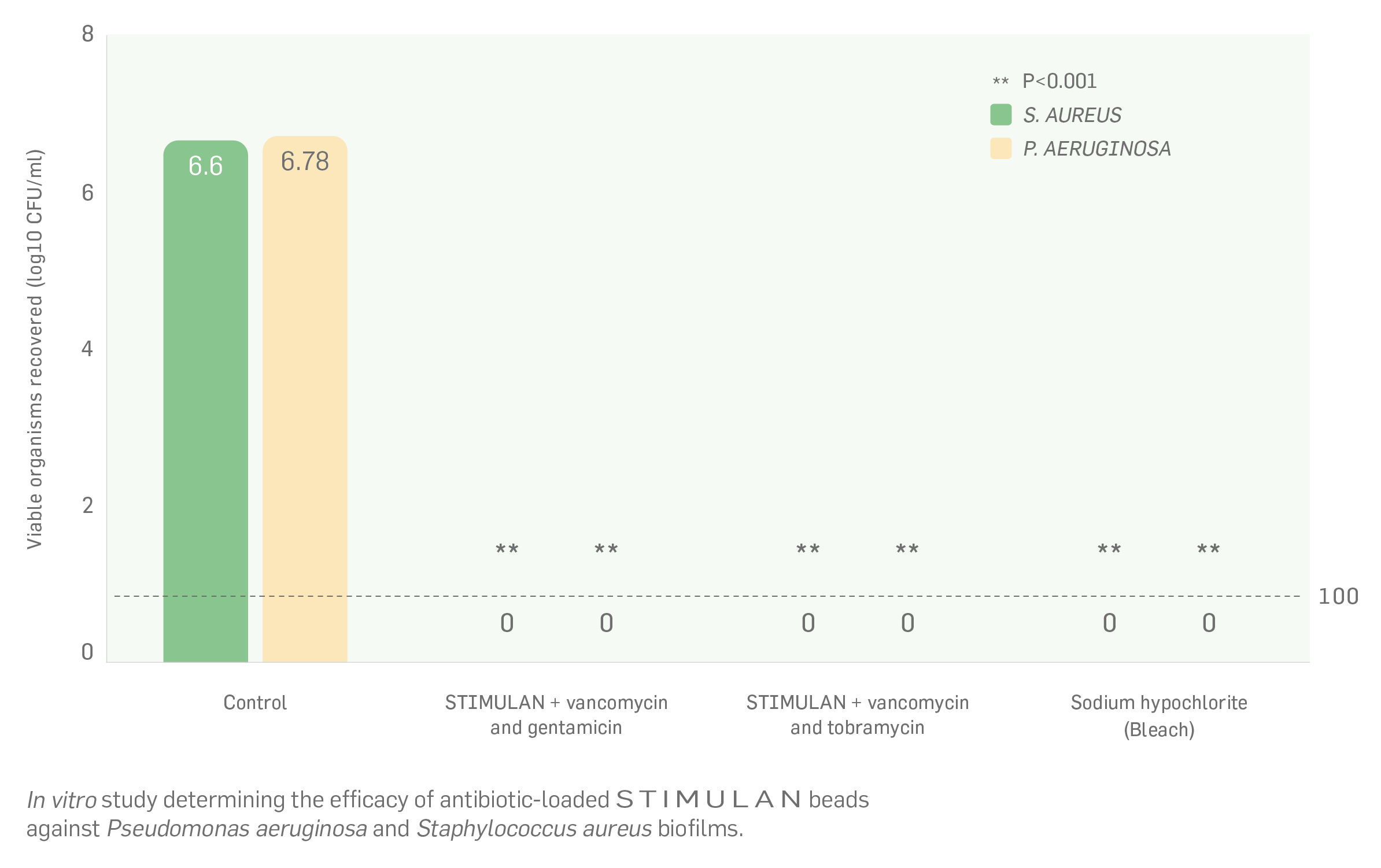
1.Biocomposites, STIMULAN Rapid Cure/Kit Instructions for Use. 2. Cooper, J.J., Method of producing surgical grade calcium sulphate; Patent. 1999. 3. Cooper, J.J., S.S. Aiken, and P.A. Laycock, Antibiotic stability in a synthetic calcium sulphate carrier for local delivery, in 32nd annual meeting of the European Bone and Joint Infection Society. 2013: Prague, Czech Republic. 4. Laycock, P., et al., In Vitro Efficacy of Antibiotics Released from Calcium Sulfate Bone Void Filler Beads. Materials, 2018. 11(11): p. 2265. 5. Delury, C., et al., Determining the Efficacy of Antibiotic-loaded Calcium Sulfate Beads against Pre-Formed Biofilms: An In Vitro Study, in ASM Microbe. 2019: San Francisco, USA. Somasundaram, K., et al., Proximal humeral fractures: the role of calcium sulphate augmentation and extended deltoid splitting approach in internal fixation using locking plates. Injury, 2013. 44(4): p. 481-7. 7. Lei, D., et al., Treatment of Distal Radius Bone Defects with Injectable Calcium Sulphate Cement., in Bone Grafting, A. Zorzi, Editor. 2012, InTech. p. 125-134. 8. Lei, D., L. Jing, and S. Yang-yong, Calcium sulfate versus calcium phosphate in treating traumatic fractures. JOURNAL OF CLINICAL REHABILITATIVE TISSUE ENGINEERING RESEARCH., 2008. 12. 9. Lei, D., Z. Ma, and X. Jing, Treatment of bone defect with injectable calcium sulfate powder in distal fractures of radius. Chinese Journal of Bone Tumor and Bone Disease, 2007. 10. Aiken, S.S., J.J. Cooper, and S. Zhou, Osseointegration of a calcium sulphate bone substitute in a large animal model, in The 5th International Congress of Chinese Orthopaedic Association. 2010: Chengdu, China. 11. Lazarou, S.A., G.B. Contodimos, and I.D. Gkegkes, Correction of alveolar cleft with calcium-based bone substitutes. J Craniofac Surg, 2011. 22(3): p. 854-7. 12. McPherson, E.J., M.V. Dipane, and S.M. Sherif, Dissolvable Antibiotic Beads in Treatment of Periprosthetic Joint Infection and Revision Arthroplasty. The Use of Synthetic Pure Calcium Sulfate (Stimulan®)Impregnated with Vancomycin & Tobramycin. Reconstructive Review, 2013. 3(1): p. 32-43. 13. McPherson, E.J., Dissolvable Antibiotic Beads in Treatment of Periprosthetic Joint Infection. The Use of Synthetic Pure Calcium Sulfate (Stimulan®) Impregnated with Vancomycin & Tobramycin, in 2nd Annual Oxford Bone Infection Conference (OBIC). 2012: Oxford, UK. 14. Gauland, C., The use of antibiotic impregnated, implanted synthetic calcium sulphate tablets in the treatment of soft tissue, vancomycin resistant, enterococcus infections, in The Symposium on Advanced Wound Care and Wound Healing Society Annual Meeting. 2011: Gaylord Texan Hotel & Convention Center, Dallas, Texas. 15. Borrelli, J., Jr., W.D. Prickett, and W.M. Ricci, Treatment of nonunions and osseous defects with bone graft and calcium sulfate. Clin Orthop Relat Res, 2003(411): p. 245-54. 16. Helgeson, M.D., et al., Antibiotic-impregnated calcium sulfate use in combat-related open fractures. Orthopedics, 2009. 32(5): p. 323. 17. McKee, M.D., et al., The use of an antibiotic-impregnated, osteoconductive, bioabsorbable bone substitute in the treatment of infected long bone defects: early results of a prospective trial. J Orthop Trauma, 2002. 16(9): p. 622-7. 18. Noor, S., et al., The use of Osteoset-T in the treatment of chronic osteomyelitis of the tibia following exogenous trauma: A review of 22 patients at a regional trauma centre. Bone & Joint Journal Orthopaedic Proceedings Supplement, 2013. 95-B(SUPP 23): p. 4. 19. Analysis of the Wear Effect 3rd Body Particulate (Bone Cement) has on UHMWPE, Accutek Testing Laboratory, Fairfield OH, K13107732-1, 2014. 20. Cowie, R.M., et al., The influence of a calcium sulphate bone void filler on the third-body damage and polyethylene wear of total knee arthroplasty. Bone Joint Res, 2019. 8(2): p. 65-72.
For indications, contraindications, warnings and precautions see Instructions for Use. Concurrent use of locally administered antibiotics may affect setting time, absorption and/or bone formation. It is the surgeon/healthcare professional’s responsibility to give due consideration to the details in the medicinal product marketing authorization in deciding whether it is appropriate for the patient under his/her care. The relevant Summary of Product Characteristics (SmPC) must be consulted. The type and dose of medicinal substance should also be assessed according to the individual patient’s clinical circumstance.
This website may include the use of STIMULAN or techniques that go beyond the current clearance / approval granted by the relevant regulatory authority. Please contact your local representative for further information.
©2024, Biocomposites, STIMULAN, Bringing Calcium to Life, Power to Transform Outcomes and DRy26 are trademarks/registered trademarks of Biocomposites Ltd. All rights reserved. No unauthorised copying, reproduction, distributing or republication is allowed unless prior written permission is granted by the owner, Biocomposites Ltd.
Patents granted: GB2367552, EP 1204599 B1, US 6780391, EP 2594231 B1, US 88883063, CN ZL201210466117.X, GB2496710, EP 3058899 B1, US 10390954
Patents pending: GB1502655.2, US 15/040075, CN 201610089710.5, GB1704688.9, EP 18275044.8, US 15/933936, CN 108619579A
MA0033R3/MA0250R3/MA0148R2/MA0074R7